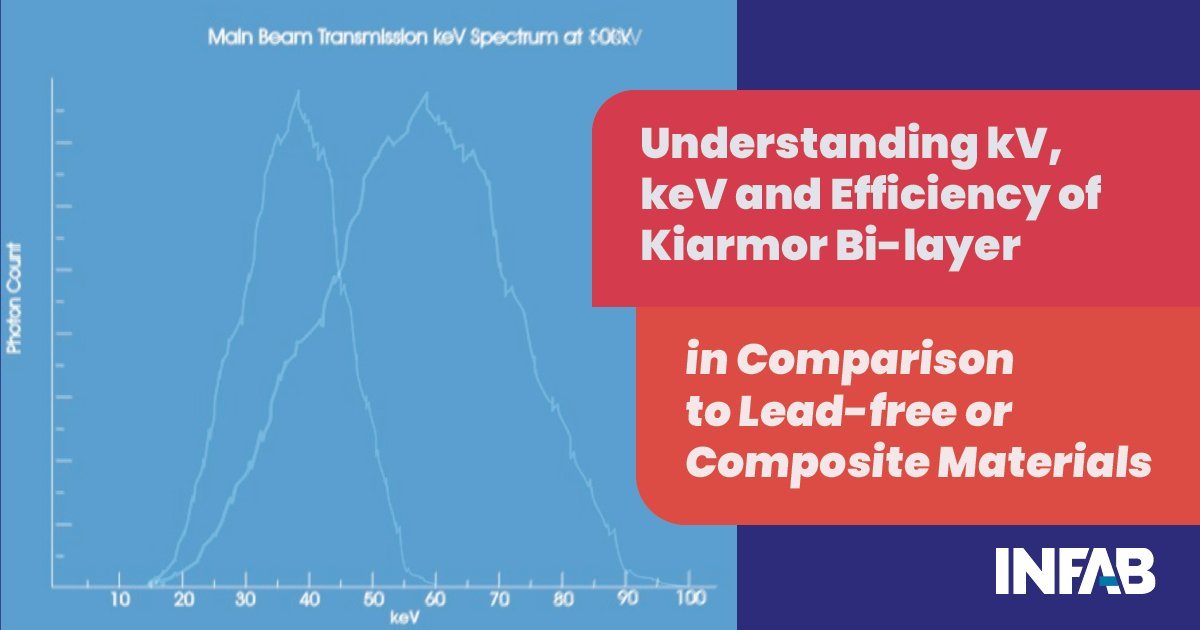
Absorbed dose is created from radiation that does not pass through the body. That is the lower ranges of keV, typically 15keV to approximately 45keV. Scatter radiation is primarily made up of this range of keV and the core material needs to be at its most efficient and effective in this range to be truly effective in protecting the user.
Given the principles above, and taking the example of fluoroscopy, which typically uses between 60kV to 70kV across the lamp to generate the X-rays, the main beam will have keV energies from 15keV to approximately 65keV. However, the scatter radiation will have energies from 15keV to approximately 45keV. This means that the vast majority of the radiation is in the absorbed dose range as it lacks the penetrating power and will stay in the body of the recipient.
Current Lead-free materials use lower-weight atomic elements, for example, Antimony, Tin, Barium, etc., to provide lighter materials that are just able to pass current test standards such as ASTM and IEC 61331-1. However, these elements have K-edges in the critical area of keV in terms of absorbed dose, which is not picked up by the test standards. Higher atomic weight elements, such as Lead or Bismuth, have K-edges beyond the keV spectrum for typical scatter radiation and do not have the same issue.
K-edges create fluorescence and a breakdown of the efficiency of these elements in terms of absorbing photon energy from an X-ray source, or the derived scatter radiation. In fact, these K-edges can actually increase the dose to the wearer of an X-ray protection apron as the fluorescence causes an increase in photon energy to the wearer of an X-ray protection apron made from these lower atomic weight elements (Fig 5). The same applies to materials using a composite (mixture) of a higher and lower atomic weight element, for example, Bismuth/Antimony, or a Lead composite material. That this occurs right next to the wearer is the worst case scenario in terms of protection against absorbed dose.